Brain Development: Conception to Age 3
The first years of life are a vital period for early brain development.
Decades of research show that the environment of a child’s earliest years can have effects that last a lifetime. The biological embedding of early experience in brain development is supported by numerous studies.1 Thanks to recent advances in studying the brain, we have a clearer understanding of how these effects are related to building early brain architecture. Neuroscientists can now identify patterns of brain activity that are associated with various types of toxic stress, such as growing up in poverty.2
Although the dangers of early toxic stress, like poverty, neglect and maltreatment, have long been recognized, we can now ‘see’ their effects using brain scanning technology. Scientists continue to do research to determine exactly how experiences affect development, and exciting advances continue to enhance intervention and prevention efforts.3
The architecture of a child’s brain is affected by early experiences.
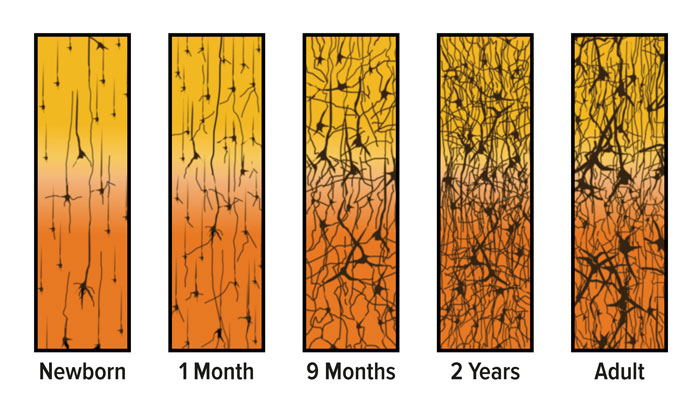
Specialized brain cells called neurons send and receive information by forming connections with one another. The connection is called a ‘synapse’. The newborn brain continues to add neurons over the first few years of life and grows at an amazing rate. It doubles in size in the first year, and by age three it reaches 80 percent of its adult volume.4-6 This growth is due mostly to neuron growth and new synapses being made.
Even more importantly, connections are formed at a faster rate during these years than at any other time. In fact, the brain creates many more connections than it needs: at age two or three, the brain has up to twice as many connections as it will have in adulthood (FIGURE 1). The experiences of a child play a big role in determining which surplus connections are gradually eliminated throughout childhood and adolescence, a process sometimes referred to as pruning.7
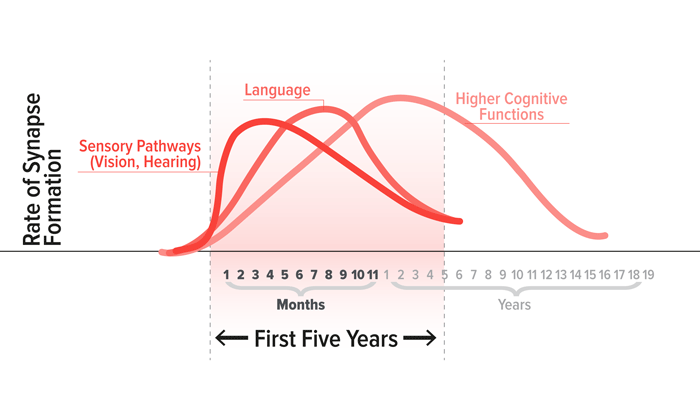
The growth and pruning of connections responsible for specific functions like vision, language or learning, occur at different rates.
Senses like hearing, vision and touch mature rapidly and are especially responsive to early external input during early infancy. The connections that are important for language development and social interactions mature over a longer period of time, but are particularly sensitive in toddlers (FIGURE 2).
For these skills, the first 3 years is the period when the brain can “capture” experience more efficiently than it will be able to later, when the pruning of unused connections is underway.7
Genetic and environmental factors work together to shape early brain development.
Although the first stages of brain development are strongly affected by genetic factors, genes do not design the brain completely.8,9 Instead, when and where genes are used is fine-tuned according to the input they receive from the environment – this happens even during pregnancy when maternal nutrition and stress can influence the early phases of brain architecture. These gene-environment relations allow for each child to adapt to their surroundings more readily and more quickly than they could if genes alone determined the brain’s wiring.10 There are two major ways that genes and environment work together to sculpt the brain.
One is through inheriting certain forms of genes that can have very different interactions with the environment.11 The second is through environmental influences that can alter the read-out of genes without changes to the genes themselves. This second process is becoming better understood thanks to recent research in a relatively new scientific field called epigenetics.
The field of epigenetics has changed our understanding of how the environment interacts with our genes and how genes interact with the environment.
Epigenetics (meaning ‘above’ genetics) is the study of enduring changes in gene activity that do not change the DNA code itself, but through chemical changes, do influence how the code is used. Many environmental factors and experiences result in chemical ‘marks’ on certain parts of genes, and these epigenetic changes can influence the activity, or ‘expression’, of the gene.12
You can think of the epigenetic processes as the software that directs the functioning of a gene’s DNA hardware. Because the development of all cells, tissues, and organs is affected by when and how specific genes are expressed, epigenetic processes can be a powerful influence on health and well-being.
Animal research shows that epigenetic changes can be long-lasting and even can be passed from one generation to the next.
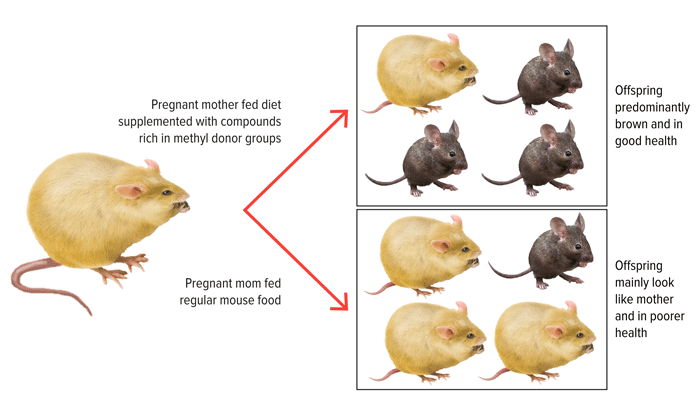
So far, much of what we know about epigenetics comes from research on animals. Numerous studies now show how genetic activity can be altered by exposure to different foods, toxins, and powerful experiences. One remarkable illustration of how potent epigenetics can be was done in genetically identical pregnant mice (like identical twins). The mothers all carried genetic information that gave rise to a yellow coat color, obesity, and vulnerability to disease. Half of the pregnant mothers received a normal diet while the other half was fed a diet high in compounds that can result in modified DNA through epigenetic mechanisms. Interestingly, the offspring of the ‘normal’ diet group resembled their mothers in coat and weight and health outcomes. The offspring from the second diet group were more likely to have brown fur, normal weight, and no increased disease risk (FIGURE 3). But like their mothers, all of the offspring in both groups had identical DNA sequences. The differences in color, weight, and health were due to differences in how genes were expressed following epigenetic changes to a specific gene. This resulted in dramatic changes in terms of how the mice appeared and even their improved health outlook. In distinction to this positive outcome, research now tells us that there are a number of external factors that occur prenatally, such as exposure to alcohol or environmental toxins that lead to negative alterations in the DNA changes and negative health outcomes.
Remarkably, the healthier female offspring eventually became pregnant and gave birth to babies that showed the same traits—brown fur, normal weight, and low disease risk—even though this third generation went back to receiving a normal diet. This experiment, and others like it, shows how influential the environment can be on epigenetics, which can have effects from one generation to the next.
Source: Illustration by Bill Day adapted from Waterland, RA., Jirtle, RL. Transposable elements: Target for early nutritional effects on epigenetic gene regulation. Molecular and Cellular Biology. 2003; 23(15):5293–5300
In another series of experiments, adult mice that had received generous amounts of licking and grooming from their mothers as pups were less anxious and had lower levels of stress hormones than those raised by mothers who showed anxious behavior and were not as nurturing. How can the differences in mother-pup interactions result in long-lasting changes in stress responses? A second phase of research was done to show that it wasn’t due to genetic differences between the different moms. To show this, pups from higher care moms were switched at birth to be with the lower care moms. The scientists also did the opposite – switch the pups from lower care moms to be with the higher care moms.
The results showed the powerful impact of early experience. Babies born to high-nurturing mothers, but switched to be raised by low-nurturing mothers grew up to express increased levels of anxious behavior similar to their foster moms. Remarkably, the mouse pups born to low-nurturing mothers but raised by high-nurturing mothers showed less anxiety. The studies also showed that a specific gene that controls stress response was expressed more highly in the mice raised by higher care moms, compared to those raised by lower care moms.13
Epigenetics is strongly related to early brain development.
We know that children’s experiences during the first years of life are strongly associated with long-term cognitive, emotional, and social outcomes.14 And we know that the quality of a child’s early experiences affects the development and function of the growing brain. But discovering how these processes occur has been challenging. The growing body of research on epigenetic processes, which are especially active early in development,15 is likely to provide new answers to how adversity threatens optimal development.
For ethical and practical reasons, it is harder to study the gene/ environment relationship in humans than in animals. Still, scientists have already found convincing evidence of epigenetic effects in human development. In one study, women who were pregnant during a severe famine tended to give birth to underweight infants. When these babies grew up and became parents themselves, they also tended to have underweight children, even though their own food intake since birth had not been affected by the famine.16 Other studies have found that childhood abuse is associated with lifelong decreased activation of a gene that protects against high levels of stress hormones.12 Recent research has found that experiences during a child’s early life can result in epigenetic changes that are apparent even when the child reaches adolescence.11,17
Studies show that high stress and low nurturing in the first stages of life impair the development of healthy brain architecture. These effects are especially dramatic in brain areas related to memory, learning, and social and emotional adjustment.13
Epigenetic research supports the importance of a preventive approach to child health and well-being.
Epigenetic processes indicate that development is remarkably flexible. But in the absence of prevention or interventions, epigenetic changes – and their effects on behavior and health – can be stable once they occur. Moreover, research tells us that such changes can be transmitted from generation to generation.18 Whether they can become permanent is not yet known, but even when the conditions that created an epigenetic mark no longer exist, it is likely to take several generations before it begins to fade.12
In other words, epigenetics makes a strong argument that prevention is the best policy approach for protecting young children from the effects of toxic stress. Early exposure to chronic stressors, such as regularly witnessing violence, caregiver neglect or abuse, poor nutrition, and other environmental hazards can have long-lasting and powerful effects on adult physical and mental well-being. Research is giving us a better understanding of epigenetic changes that occur due to early adverse experiences, which will lead to the development of more effective intervention and prevention programs to protect young children from adverse experiences in the first years of life.19,20
- Hertzman C. Commentary on the symposium: biological embedding, life course development, and the emergence of a new science. Annual Review of Public Health. 2013; 34:1–5.
- Lipina SJ, Colombo JA. Poverty and Brain Development During Childhood: An Approach From Cognitive Psychology and Neuroscience. Washington, DC: American Psychological Association; 2009.
- Sheridan MA, Fox NA, Zeanah CH, McLaughlin KA, Nelson CA. Variation in neural development as a result of exposure to institutionalization early in childhood. Proc Natl Acad Sci U S A. 2012; 109(32):12927–32.
- Gilmore JH, Lin W, Prasatwa MW, et al. Regional gray matter growth, sexual dimorphism, and cerebral asymmetry in the neonatal brain. Journal of Neuroscience. 2007; 27(6):1255–1260.
- Nowakowski RS. Stable neuron numbers from cradle to grave. Proceedings of the National Academy of Sciences of the United States of America. 2006; 103(33):12219–12220.
- Rakic, P. No more cortical neurons for you. Science. 2006; 313:928–929.
- Huttenlocher P. Neural Plasticity: The Effects of the Environment on the Development of the Cerebral Cortex. Harvard University Press; 2002.
- Kagan J, Herschkowitz N, Herschkowitz E. A Young Mind in a Growing Brain. Mahwah, NJ: Lawrence Erlbaum Associates; 2005.
- Elman JL, Bates EA, Johnson MH, et al. Rethinking Innateness: A Connectionist Perspective on Development. Cambridge, MA: MIT Press; 1996.
- Pascual-Leone A, Amedi A, Fregni F, et al. The plastic human brain cortex. Annual Review of Neuroscience. 2005; 28:377–401.
- Feder A, Nestler EJ, Charney DS. Psychobiology and molecular genetics of resilience. National Review of Neuroscience. 2009; 10(6):446–57.
- McGowan PO, Szyf M. The epigenetics of social adversity in early life: Implications for mental health outcomes. Neurobiology of Disease . 2010; 39(1):66–72.
- Meaney, M. Epigenetics and the biological definition of gene x environment interactions. Child Development. 2010; 81(1), 41–79.
- Duncan GJ, Ziol-Guest KM, Kalil A. Early childhood poverty and adult attainment, behavior, and health. Child Development. 2010; 81:306–325.
- Fagiolini M, Jensen CL, Champagne FA. Epigenetic influences on brain development and plasticity. Current Opinion in Neurobiology. 2009; 19:1–6.
- Heijmans BT, Tobi EW, Stein AD, Putter H, Blauw GJ, Susser ES, Slagboom PE, Lumey LH. Persistent epigenetic differences associated with prenatal exposure to famine in humans. Proc Natl Acad Sci U S A. 2008; 105(44):17046–9.
- Gleason G, Zupan B, Toth M. Maternal genetic mutations as gestational and early life influences in producing psychiatric disease-like phenotypes in mice. Front Psychiatry. 2011; 2,25:1–10.
- Francis DD. Conceptualizing child health disparities: a role for developmental neurogenomics. Pediatrics. 2009; 124:S196–S202.
- Essex MJ, Boyce WT, Hertzman C, et al. Epigenetic vestiges of early developmental adversity: Childhood stress exposure and DNA methylation in adolescence. Child Development. 2013; 84:58–75.
- Shonkoff JP, Levitt P. Neuroscience and the future of early childhood policy: Moving from why to what and how. Neuron. 2010; 67:689–691.